GuardCap™ ready-to-fill, functional capsules for colonic delivery of nutraceuticals and food supplements
Leveraging the power of EUDRAGUARD® biotic for colonic delivery and streamlined nutraceutical development
Bringing a dietary supplement to market requires rapid development. This can be challenging when the health ingredients like vitamins, probiotics and prebiotics are sensitive to gastric environments. Gastric acid, bile acid and enzymes can break down sensitive ingedients before their nutritional and health benefits can be released. Mechanical stress, moisture and heat that is experience during the coating process for capsules also needs to be considered when developing dietary supplements. Evonik has created a ready-to-fill hard capsule, coated with the proven and well-known EUDRAGUARD® biotic functional coating for nutraceuticals. This unique capsule is suitable for colon delivery, increasing the viability of ingredients like probiotics and prebiotics which contain sensitive bacteria. It also saves costs during production development by reducing the number of steps required.
Challenges in developing colonic delivered nutraceuticals
Ensuring that sensitive health ingredients can be released at the right time and place can be a challenge for all dietary supplement developers. Delayed release offers many benefits including protecting ingredients that may degrade in the digestive environment of the stomach and releasing ingredients directly where they can be absorbed. Many health ingredients require protection from gastric acid, bile acid and enzymes during digestion. This is especially true for probiotic bacteria where 60 percent of strains are killed in the gastric environment before they reach the intestine. Many probiotic bacteria strains and health ingredients are also sensitive to moisture, heat, or mechanical stress – all of which can occur during coating processes.
Rapid nutraceutical development demands efficiency
Nutraceutical development is a fast-moving business. Lifecycles for supplements can be short and fast-changing consumer requirements, coupled with swift scientific development mean that new products have to be launched frequently and developed fast. One way of reducing the development timeline is by streamlining time-consuming steps. Testing different coatings for stability or scale-up trials for coating processes, for example, may hold up your ambitious timeline.
Securing the supply for nutraceutical development
Having your own coating capabilities is not a given for many nutraceutical companies. This means that coating must be carried out by a contract manufacturing organization (CMO). Using a CMO can increase the complexity of the supply chain and prolong the production time for a supplement. This is relevant for developers working on probiotic supplements, where some probiotic strains such as spore formers are not desirable for CMOs due to the risk of cross-contamination.
GuardCap™ for targeted release of nutraceuticals
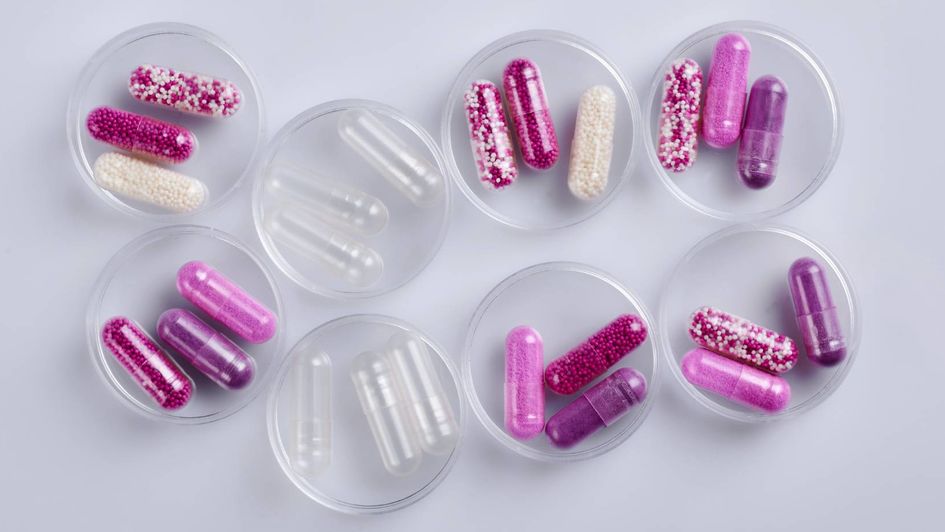
GuardCap™ is based on an HPMC capsule that is coated with an EUDRAGUARD® biotic-based formulation. The coating is applied across the entire surface including the gap between body and cap. This guarantees the protection of sensitive ingredients from gastric acid, bile acid and enzymes and ensures targeted release in the colon at acidic pH ranges.
The capsules are delivered in a pre-locked status and ready to fill. They are easy to open, fill and close and are fully compatible with conventional capsule filling machines. They can also be filled by hand during the test phase.
Suitable for a range of fillings such as powders, pellets, granules, and even liquids, our ready-to-fill capsules ensure fast development of new dietary supplements. This helps you reduce complexity in your supply chain.
Other key benefits include:
- Available in different capsule sizes
- Batch size flexibility for your final product
- Easy-to-swallow
- Reliable dissolution and disintegration
- Quality compliance with regulations for dietary / food supplements
- Storage-stable capsule
- No additional sealing or banding needed
- Free from formulation: Solvent-fee, TiO2-free, talc-free
We understand that your consumers have the highest expectations when it comes to nutrition and health. Find out more about how you can use GuardCap™ in your next project.
Are you developing a dietary supplement with sensitive ingredients? Would you like to get more information on how extended release is possible for your next oral supplement? Or perhaps you are working on a new probiotic supplement? Find out how GuardCap™ can help.