Your Preferred 3D Printing Partner for Medical Polymers And Other Device Solutions
An integrated portfolio of 3D printing, implantable medical polymers backed by advanced application services
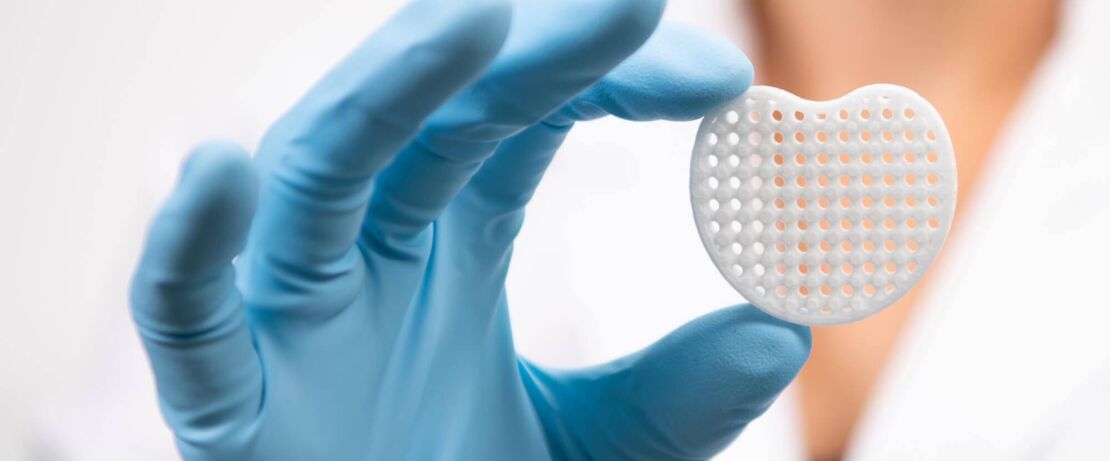
Evonik has brought together a broad, flexible portfolio for 3D printing including medical polymers and advanced application services to enable the rapid development of bioresorbable implant devices for patient-specific solutions. As the world’s first commercial GMP and ISO 13845 certified supplier of biodegradable medical materials for 3D printing including powders, filaments and granules, we can provide standard and custom grades for use with all key additive manufacturing technologies. In addition to our RESOMER® line of medical polymers designed for 3D printing, customers can leverage our technical knowledge and global network of application labs to help reduce project risk and accelerate their development and scale-up timelines. Get in touch with us to discover how we can be of assistance for your next 3D printing project requiring medical grade polymers.
A complete range of implantable biodegradable medical polymers for all 3D printing technologies
Our portfolio of biodegradable medical polymers for 3D printing has been developed to provide medical device companies with the flexibility they require to create patient-specific parts for use across a range of application areas including cranioplasty, craniomaxillofacial and skull plates, spinal and large-bone cages, lung implants and bone scaffolds for use in orthopaedic, spine, dental and regenerative medicine applications. Based upon specific application requirements, we can provide medical polymers for a use with 3D printing technologies including Fused Filament Fabrication (FFF, and also known as Fused Deposition Modeling), Selective Laser Sintering (SLS), Bioplotter and Extrusion-based systems. This portfolio includes GMP powders for SLS applications, GMP filaments for Fused Filament Fabrication applications, and GMP polymer granulates for extrusion-based 3D printing technologies such as freeformers. In addition to being the only supplier with such a unique range of functional medical materials for 3D printing, we have a history of safety and biocompatibility that exceeds more than 30 years of commercial use.

This whitepaper reviews the suitability of bioresorbable polymers for use with Selective Laser Sintering (SLS), Fused Filament Fabrication (FFF), extrusion-based Freeform printing, and Bioplotting. These technologies represent the four of the most common 3D printing systems used today to develop bioresorbable implantable devices suitable for human use.
RESOMER® PrintPowder: a biodegradable powder-based medical polymer for 3D printing with Selective Laser Sintering technologies
RESOMER® PrintPowder is a free-flowing powder-based medical polymer with tight product specifications enabling higher resolution 3D printing with Selective Laser Sintering technologies. Featuring an optimized particle size distribution for efficient processability, RESOMER® PrintPowder ensures a consistent flow of high-quality powder during the 3D printing process to ensure it is assembled to precise geometrical requirements. Mechanical properties and degradation characteristics of devices utilizing RESOMER® PrintPowder can also be precisely engineered to meet key application requirements such as strength, durability and the prevention of stress shielding. We can provide customized RESOMER® Select powders to address your specific application needs.
RESOMER® Filament: a biodegradable filament-based, high resolution 3D printing medical polymer with Fused Filament Fabrication technologies
Our line of RESOMER® Filaments for 3D printing can support the development of higher resolution medical polymers printed using Fused Filament Fabrication technologies. Poly (L-lactide) or PLLA, Poly (L-lactide-co-glycolide) or PLGA, Poly (caprolactone) or PCL, and Polydioxanone or PDO-based grades are available with tight specifications including a 1.75 mm diameter size and precise processing temperatures for the nozzle, bed and chamber. All filaments for 3D printing are provided coiled in a high-quality spool package size of 50 grams. Mechanical properties such as strength and elongation can be tailored to the target application, with degradation times ranging from less than six months to more than three years. We can also provide customized RESOMER® Select filaments to address your specific application needs.
Speed to market: Our 3D printing services enable you for faster product development and scale-up of your device
Evonik’s advanced powder preparation capabilities can facilitate the GMP supply of ISO 13485-certified powder-based medical polymer formulations for 3D printing suitable for both development and commercial use. Other application services include the rapid printing and evaluation of feasibility samples, non-GMP prototyping, process development support, analytical characterization, mechanical testing and degradation studies. With application technology labs and competence centers in the U.S., Germany and China, we have technical expertise and additive manufacturing capabilities to provide reliable and responsive support until the finished 3D printed device part utilizing our medical polymers is in your hands.

Looking for information compiled in one document? Our brochures offer an overview of relevant details in just a few pages.






